
Custom batteries for medical devices

For over 30 years, we have been supporting custom battery pack projects for demanding applications, including the medical sector and related uses (veterinary and cosmetics).
Our approach has a simple goal: to secure your project from the outset using your specifications, by aligning energy performance, mechanical/electrical integration, risk management and the documentary traceability expected in this type of environment.
Medical sector
Non-negotiable requirements (safety, reliability, traceability)
In a medical device, the battery determines the availability of the device, patient/user safety, maintenance and documentation control.
Based on your constraints, we define: chemistry, mechanics, electronics and charging strategy.
From experience, we have found that two medical devices that appear similar may require very different battery architectures depending on the following criteria:
-
Mission profile
(autonomy, peak power, cycles)
-
Environmental conditions
(temperature, shocks)
-
Integration constraints
(space requirements, connectivity, ergonomics)
-
Target market
(country, transport, applicable regulatory requirements)
Compliance & quality framework:
Securing the project from the design stage onwards
ISO 13485 Design and manufacture of medical devices

IEC 60601 Electrical safety and performance of medical devices
Our architectural and design choices are intended to facilitate the integration of batteries into devices subject to IEC 60601 standards.
UN38.3 Lithium transport (testing & shipping)
EU / International Regulatory framework for batteries
- Compliance guidelines (product lifecycle, EU requirements, etc.): Regulations – Batteries and accumulators
- Depending on the market: IEC and UL standards (e.g. UL 2054) may be relevant to certain testing and qualification strategies.
Technological choices:
selecting the right chemistry for the right application
We work with several technologies and select the architecture based on the specifications:

Lead Acid
Simplicity, robustness, cost; typically 6V or 12V configurations depending on requirements.

Lithium
Energy density, various formats, charging strategy and associated electronics.

Nickel-Metal Hydride
Balance between durability & maintenance requirements, depending on specific needs & constraints.
Custom medical battery design:
from specifications to market launch

Architectural review and scoping of constraints
We begin by analysing your specifications for the design of a medical battery: available form factor (volume/mass), interfaces, usage profile (energy/power, peaks), power consumption, charging strategy, operating environment and target markets.
Objective: to identify the key design choices (chemistry/cells, series-parallel architecture, protection/measurement systems, mechanical/electrical interfaces) and establish a realistic roadmap (performance, validation, industrialisation) for your medical battery.

Validation with your equipment
We design and manufacture prototypes, then work with you to organise the validation phase on your equipment.
We generally offer two prototypes:
• 1 ENIX prototype: intended for configuration monitoring, medical battery design iterations and the finalisation of the specification document.
• 1 Client prototype (testing): intended for integration and testing in real conditions by your teams, directly in your application and according to your operating scenarios.
The preliminary study and prototyping phases allow us to validate the solution against the specifications (energy/power, integration, interfaces, behaviour in use) and to adjust the architecture before your bespoke medical battery goes into production.

requirements
The compliance of a custom medical battery is defined according to the product scope and the countries in which it will be marketed. From the scoping phase onwards, we integrate the applicable requirements in order to secure the architecture (pack + associated electronics), documentation and market launch trajectory.
Depending on your market, the framework includes UN38.3 (transport) and, where applicable, CE marking requirements, with the associated safety and sustainability expectations. We also assist you in preparing the necessary elements for export compliance (non-EU countries) in order to facilitate access to your international markets.
Additional standards (e.g. UL, local requirements) are assessed on a case-by-case basis according to marketing objectives and integration constraints.

From prototype to consistent quality at target volume
Industrialisation aims for controlled production, with consistent quality, at the target volume:
definition → qualification → production.
We manage the scale-up process, from small-batch production to industrial-scale manufacturing, with a controlled production process, ensuring a custom medical battery that remains reliable over time.

Validation under real conditions
The validation of the pack is carried out by your teams, under real-world conditions and within your application, in line with your usage scenarios (profile, environment, cycles).
We work with you to define the validation plan and acceptance criteria to ensure that the design is finalised and your custom medical battery can proceed to production.
Please note: the testing phase can sometimes take several months, depending on the application and usage cycles.

Safety:
protection systems, monitoring and control of the electronics (BMS / PCM)
The design incorporates the appropriate level of protection and monitoring right from the preliminary design stage:
- electrical/thermal protection,
- shutdown strategy, safety devices,
- communication protocols,
- and, where necessary, load balancing and condition monitoring (SOC/SOH).
Partners & support teams:
ensuring availability and technical consistency
Enix Power Solutions draws on a network of recognised cell and battery manufacturers and suppliers, selected based on availability, the required formats and compliance with the specifications. These include Saft, Tadiran, Energizer, Ultralife and Panasonic, depending on the project.
Our role: to help you arrive at a robust solution by combining cell selection, pack architecture and integration constraints.
Our aim is to mitigate project risk through:
- selection criteria consistent with the intended use and the required safety level,
- qualifiable alternatives in the event of changes to the supply chain,
- documentation and traceability tailored to the project.
Medical applications:
ensuring availability and technical consistency
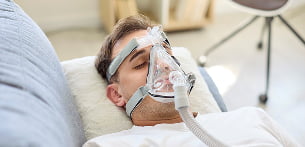
Respiratory equipment
Depending on requirements, 12V lead-acid battery systems or lithium-based solutions tailored to specific needs are commonly used, including Li-SOCl₂…
to facilitate replacement without need for recharging. Working in partnership with manufacturers such as SAFT, Tadiran and Yuasa, Enix Power Solutions has supported projects involving respiratory equipment, including during the COVID-19 pandemic, whilst taking into account the requirements for uninterrupted power supply and availability.
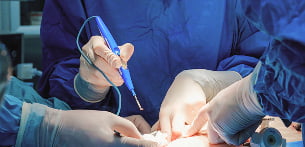
Surgical instruments
As they generally require high-power lithium-ion cells, surgical power tools must be designed with a clear focus on ergonomics, safety and repeatability of use (power, cycle life, thermal performance).
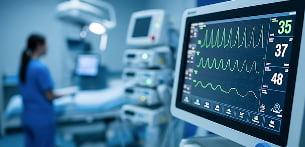
Patient monitors
For patient monitors, we offer standard ‘ready-to-use’ batteries and bespoke battery packs, tailored to the required battery life, mobile use and charging/maintenance requirements.
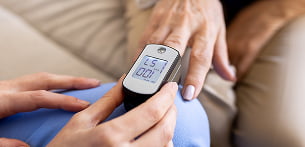
Diagnostic tools
These diagnostic tools enable practitioners to work outside the clinic; the battery must therefore prioritise portability, genuine battery life and reliable performance (charging, cycle life, durability).

Medical imaging
Requirements vary greatly depending on the equipment, but battery design must take into account safety, availability and system integration from the outset.
Portable medical devices
Availability is also crucial in hospital settings. This emphasis on mobility also applies to mobility aids, where weight, battery life and durability are key factors.

Robotic orthosis
Robotic orthoses place significant demands on weight, size, power and durability in real-world use, with particular attention paid to safety and cycle times.
Are you developing a medical device? Let's discuss energy architecture.
Once we have received your documentation, an Enix Power Solutions engineer will contact you to discuss and finalise the specifications (intended use, integration constraints, environment, safety and traceability requirements).
This discussion enables us to confirm the key points and begin the architectural design phase, ensuring the project gets off to a solid start based on a clear and shared technical understanding.
Advantages of using Enix Power Solutions for your project:
- Bespoke solutions built to your specification
- Design & Characterisation
Speak to a member of our OEM specialist team about custom drone batteries
- Contact Form
- UK: +44 (0) 121 506 8610
- Custom Battery Pack Enquiry










